How do they fight cancer?
Context
The idea of immunotherapy or using our own immune system against cancer has been around for more than 60 years. At the time, the absence of techniques for culturing and manipulating cells made it impossible to harness the power of immune cells. Decades later, scientists were able to design and produce antibodies targeting specific molecules on the surface of cancer cells. Such therapies were only approved for patients in the late 90s.
CAR-T, a revolutionary cell therapy for cancer has been recently approved by the FDA. But what is it about?
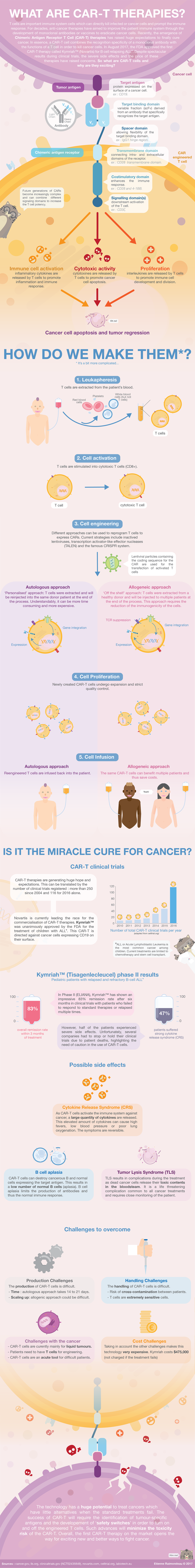
More recently, a radical new approach has been allowed by genome editing enzymes such as TALENs and the famous CRISPR proteins : to engineer immune cells to target cancer. Lymphocytes are the specialised soldiers in the immune system army, in particular, lymphocytes T (T cells) are able to find and kill abnormal cells as well as coordinate attacks by the other immune cells. They thus constitute a perfect candidate for scientists to engineer an immune response against cancer. Antigen receptors are ‘molecular scanners’ at the surface of the T cells enabling the detection of abnormal cells. These can be modified via genome editing to force T cells to bind and destroy cancer cells. This new generation of immunotherapies has been showing incredible positive results during clinical trials which led to the approval of the first CAR-T few months ago.
Let’s explore what CAR-T are, how we make them: and why this is an exciting new way to fight cancer:
References
- Cancer.gov, “CAR T Cells: Engineering Patients’ Immune Cells to Treat Their Cancers”
- Leukemia & Lymphoma Society, “Chimeric Antigen Receptor (CAR) T-cell Therapy”
- Clinicaltrial.gov, Kymriah Phase II results
- CellTrial.info, “Number of CAR cell therapy trials worldwide”
- Labiotech.eu, “A Cure for Cancer? How CAR-T Therapy is Revolutionizing Oncology”
Thanks for reading! Let us know what you think!